|
Diagnosis
 Lyme
disease should be considered when any of the following
conditions is present: monoarticular or oligoarticular
arthritis, aseptic meningitis, cranial and peripheral
neuritis, and primary cardiac conduction abnormalities.
Bell's Palsy, once always considered to be "idiopathic" may
be a symptom of Lyme disease. History of exposure to an
endemic area within a few weeks of onset of illness should
be sought, but inquiry as to tick bites is not particularly
helpful because attachment of the Ixodes tick is usually
unnoticed and non-vector ticks are common in endemic areas.
Because Lyme disease in its early stages can mimic many
other conditions, accurate diagnosis is important before
committing the patient to a prolonged course of
antibiotics. Lyme
disease should be considered when any of the following
conditions is present: monoarticular or oligoarticular
arthritis, aseptic meningitis, cranial and peripheral
neuritis, and primary cardiac conduction abnormalities.
Bell's Palsy, once always considered to be "idiopathic" may
be a symptom of Lyme disease. History of exposure to an
endemic area within a few weeks of onset of illness should
be sought, but inquiry as to tick bites is not particularly
helpful because attachment of the Ixodes tick is usually
unnoticed and non-vector ticks are common in endemic areas.
Because Lyme disease in its early stages can mimic many
other conditions, accurate diagnosis is important before
committing the patient to a prolonged course of
antibiotics.
 The
diagnosis of Lyme Disease is based primarily on clinical
findings. Treatment without absolute confirmation of the
disease is often appropriate if the signs and symptoms fit.
However, good judgment is necessary, since patients are
often very fearful and may insist on being tested for
infection or treated for Lyme disease when little evidence
of it exists. Serologic testing for antibodies may provide
supportive information in patients in endemic areas with
findings suggesting chronic or later stages of disseminated
Lyme Disease. A positive antibody result in the face of
vague symptoms can add to the problem. A positive antibody
test may signal a previous infection that has resolved
without complications. Physicians should explain to these
patients the differences in "background" seropositivity in
various geographic locations and the drawbacks of
instituting unnecessary treatment for inactive disease. A
newer test in which the DNA of the Lyme organism can be
detected and amplified by the polymerase chain reaction
(PCR) for identification and quantification. If the patient
has a classic EM lesion or provides an unambiguous history
of this rash, in association with typical symptoms, the
diagnosis is confirmed. Atypical rashes, a vague history, or
no rash at all do not exclude the diagnosis, but then
additional evidence is obviously necessary. If the illness
involves more than one system with a compatible dysfunction,
the diagnosis is more likely if other conditions can be
excluded. The unusual skin lesion acrodermatitis chronica
atrophicans is also pathognomonic. The
diagnosis of Lyme Disease is based primarily on clinical
findings. Treatment without absolute confirmation of the
disease is often appropriate if the signs and symptoms fit.
However, good judgment is necessary, since patients are
often very fearful and may insist on being tested for
infection or treated for Lyme disease when little evidence
of it exists. Serologic testing for antibodies may provide
supportive information in patients in endemic areas with
findings suggesting chronic or later stages of disseminated
Lyme Disease. A positive antibody result in the face of
vague symptoms can add to the problem. A positive antibody
test may signal a previous infection that has resolved
without complications. Physicians should explain to these
patients the differences in "background" seropositivity in
various geographic locations and the drawbacks of
instituting unnecessary treatment for inactive disease. A
newer test in which the DNA of the Lyme organism can be
detected and amplified by the polymerase chain reaction
(PCR) for identification and quantification. If the patient
has a classic EM lesion or provides an unambiguous history
of this rash, in association with typical symptoms, the
diagnosis is confirmed. Atypical rashes, a vague history, or
no rash at all do not exclude the diagnosis, but then
additional evidence is obviously necessary. If the illness
involves more than one system with a compatible dysfunction,
the diagnosis is more likely if other conditions can be
excluded. The unusual skin lesion acrodermatitis chronica
atrophicans is also pathognomonic.
In cases in which clinical
features are equivocal, the following tests may be
approriate. 1) Lyme Western Blot, IgG, IgM; 2) Lyme ELISA;
3) Lyme DNA by PCR (skin, blood, synovial fluid,
cererbrospinal fluid, urine). Tests for other tick-borne
diseases with similar symptoms may need to be excluded with
Babesia DNA by PCR, Ehrlichia HGE and HME Western Blot
antibody testing. Other disease that may have similar vague
symptoms include Bartonella, Tularemia, Brucella, Mycoplasma
and Chlamdia.
 Lyme
disease has become a controversial and politicized disease.
This has occurred because of the seriousness of the disease
and the degree of disability it can produce. This is
especially true if there is a delay in diagnosis and
treatment. There is no doubt that much needs to be done to
prevent such complications, and to treat such sufferers
effectively. There are excesses on both sides of the
problem; those who diagnose Lyme disease too frequently and
treat when not necessary, and those who never treat without
unequivocal proof of Lyme disease. Cloning of the DNA for
several of B. burgdorferi antigens has been
accomplished. Further development of methods utilizing this
technology should allow rapid detection of the presence of
the organism itself, and would confirm difficult cases in
the future. Those interested in following new developments
in Lyme disease may want to subscribe to the Internet
newsgroup sci.med.diseases.lyme, or
participate in other Internet sites dedicated to Lyme
disease and its victims. Lyme
disease has become a controversial and politicized disease.
This has occurred because of the seriousness of the disease
and the degree of disability it can produce. This is
especially true if there is a delay in diagnosis and
treatment. There is no doubt that much needs to be done to
prevent such complications, and to treat such sufferers
effectively. There are excesses on both sides of the
problem; those who diagnose Lyme disease too frequently and
treat when not necessary, and those who never treat without
unequivocal proof of Lyme disease. Cloning of the DNA for
several of B. burgdorferi antigens has been
accomplished. Further development of methods utilizing this
technology should allow rapid detection of the presence of
the organism itself, and would confirm difficult cases in
the future. Those interested in following new developments
in Lyme disease may want to subscribe to the Internet
newsgroup sci.med.diseases.lyme, or
participate in other Internet sites dedicated to Lyme
disease and its victims.
 Isolation
of the organism B. burgdorferi is a difficult and
low-yield procedure. Direct examination of tissues rarely
reveals the spirochetes, but has been reported in brain
biopsy in a patient with subacute
encephalitis. Isolation
of the organism B. burgdorferi is a difficult and
low-yield procedure. Direct examination of tissues rarely
reveals the spirochetes, but has been reported in brain
biopsy in a patient with subacute
encephalitis.
 Serologic
analysis is the laboratory diagnostic method. Both
immunofluorescent and enzyme-linked immunosorbent assay
techniques are widely available although not standardized.
IgM response peaks in the third to sixth week of the
disease, followed by IgG and IgA responses. The IgM titer
normalizes after recovery, while the IgG titer could persist
over years or decades. Serologic tests are frequently
negative in early disease, and early antibiotic therapy may
prevent or modify the antibody response. Also, some patients
become seronegative with antibiotic therapy even though the
disease remains active. Cross reactivity exists between the
spirochete of syphilis (Treponema pallidum), the
Borrelia of relapsing fever, and B. burgdorferi.
Syphilis can be differentiated by the RPR test, but
confusion with relapsing fever might be a diagnostic problem
in areas in which both diseases are
endemic. Serologic
analysis is the laboratory diagnostic method. Both
immunofluorescent and enzyme-linked immunosorbent assay
techniques are widely available although not standardized.
IgM response peaks in the third to sixth week of the
disease, followed by IgG and IgA responses. The IgM titer
normalizes after recovery, while the IgG titer could persist
over years or decades. Serologic tests are frequently
negative in early disease, and early antibiotic therapy may
prevent or modify the antibody response. Also, some patients
become seronegative with antibiotic therapy even though the
disease remains active. Cross reactivity exists between the
spirochete of syphilis (Treponema pallidum), the
Borrelia of relapsing fever, and B. burgdorferi.
Syphilis can be differentiated by the RPR test, but
confusion with relapsing fever might be a diagnostic problem
in areas in which both diseases are
endemic.
 The
current immunodiagnostic tests for Lyme disease have to be
interpreted with caution mainly because of low sensitivity
in some clinical situations. There are reports that
serologic tests my be falsely negative in patients partially
treated with an antibiotic early in the disease. Lyme titers
may be elevated in patients whose symptoms are from other
conditions since asymptomatic infection with B.
burgdorferi also occurs. The
current immunodiagnostic tests for Lyme disease have to be
interpreted with caution mainly because of low sensitivity
in some clinical situations. There are reports that
serologic tests my be falsely negative in patients partially
treated with an antibiotic early in the disease. Lyme titers
may be elevated in patients whose symptoms are from other
conditions since asymptomatic infection with B.
burgdorferi also occurs.
 In
early Lyme disease patients may have elevations of the
sedimentation rate, and moderate elevations of transaminase
are not unusual. Tests for Rheumatoid Factor and Antinuclear
Antibodies are usually negative. Synovial fluid is usually
Type II (i.e., 2000 to 25,000 WBCs/.L), but occasional
patients have very high cell counts (>100,000 WBCs/.L). A
mild lymphocytosis may be present in the cerebrospinal fluid
when neurologic abnormalities are present. Antibodies to
B. burgdorferi may be present in synovial fluid and
cerebrospinal fluid. The cerebrospinal fluid may show
oligoclonal bands. In
early Lyme disease patients may have elevations of the
sedimentation rate, and moderate elevations of transaminase
are not unusual. Tests for Rheumatoid Factor and Antinuclear
Antibodies are usually negative. Synovial fluid is usually
Type II (i.e., 2000 to 25,000 WBCs/.L), but occasional
patients have very high cell counts (>100,000 WBCs/.L). A
mild lymphocytosis may be present in the cerebrospinal fluid
when neurologic abnormalities are present. Antibodies to
B. burgdorferi may be present in synovial fluid and
cerebrospinal fluid. The cerebrospinal fluid may show
oligoclonal bands.
Management
 Lyme
disease is usually a relatively self-limited illness. At
times, however, it is a debilitating, chronic disease. Death
from Lyme disease is rare. Lyme
disease is usually a relatively self-limited illness. At
times, however, it is a debilitating, chronic disease. Death
from Lyme disease is rare.
 Early
disease is best treated with Doxycycline orally in a dose of
100 mg two or three times a day for 21 to 30 days, or with
Amoxicillin 500 - 1000 mg three times a day for 21 to 30
days. Benemid 500 mg three times a day is sometimes given
along with amoxicillin to give a higher and more prolonged
blood level of the antibiotic. Another alternate oral
therapy is Tetracycline, 500 mg 4 times a day for 21 to 30
days. These oral regimens can also be used in the presence
of facial palsy, or mild cardiac abnormalities. Early
arthritis, synovitis, and the skin lesions of acrodermatitis
chronica atrophicans and lymphocytoma are treated with this
oral antibiotic dose for 30 days. Treatment regimens of more
than 30 days have not yet been evaluated and therefore are
not currently indicated. In children who are allergic to
penicillin, Erythromycin is used, but this antibiotic does
not reliably prevent sequelae. Prophylactic antibiotic
treatment for a tick bite is not advisable. Early
disease is best treated with Doxycycline orally in a dose of
100 mg two or three times a day for 21 to 30 days, or with
Amoxicillin 500 - 1000 mg three times a day for 21 to 30
days. Benemid 500 mg three times a day is sometimes given
along with amoxicillin to give a higher and more prolonged
blood level of the antibiotic. Another alternate oral
therapy is Tetracycline, 500 mg 4 times a day for 21 to 30
days. These oral regimens can also be used in the presence
of facial palsy, or mild cardiac abnormalities. Early
arthritis, synovitis, and the skin lesions of acrodermatitis
chronica atrophicans and lymphocytoma are treated with this
oral antibiotic dose for 30 days. Treatment regimens of more
than 30 days have not yet been evaluated and therefore are
not currently indicated. In children who are allergic to
penicillin, Erythromycin is used, but this antibiotic does
not reliably prevent sequelae. Prophylactic antibiotic
treatment for a tick bite is not advisable.
 Lyme
disease that is manifest as serious central nervous system
disease, high-degree atrioventricular block, or persistent,
proliferative synovitis is treated with intravenous
antibiotics: Cefotaxime, 3 Grams every 12 hours for 14 to 21
days; or Ceftriaxone, 2 Grams daily for 14 - 21 days; or
Penicillin G, 20 million units in divided doses daily for 14
- 21 days. Intravenous antibiotic therapy as described is
also used, in a 14 day course, for a treatment failure with
oral antibiotics. Some sequelae respond well to antibiotic
therapy while others, such as chronic arthritis or advanced
central nervous system disease may not. This fact should be
considered before subjecting patients to repeated courses of
adequate intravenous therapy. Persistent symptoms,
especially fibrositis and fibromyalgia, may be caused by a
process that is no longer antibiotic-sensitive. Special care
in the management of so-called chronic Lyme disease. is
crucial to prevent prolonged or unending courses of
antibiotics for such noninfectious
problems. Lyme
disease that is manifest as serious central nervous system
disease, high-degree atrioventricular block, or persistent,
proliferative synovitis is treated with intravenous
antibiotics: Cefotaxime, 3 Grams every 12 hours for 14 to 21
days; or Ceftriaxone, 2 Grams daily for 14 - 21 days; or
Penicillin G, 20 million units in divided doses daily for 14
- 21 days. Intravenous antibiotic therapy as described is
also used, in a 14 day course, for a treatment failure with
oral antibiotics. Some sequelae respond well to antibiotic
therapy while others, such as chronic arthritis or advanced
central nervous system disease may not. This fact should be
considered before subjecting patients to repeated courses of
adequate intravenous therapy. Persistent symptoms,
especially fibrositis and fibromyalgia, may be caused by a
process that is no longer antibiotic-sensitive. Special care
in the management of so-called chronic Lyme disease. is
crucial to prevent prolonged or unending courses of
antibiotics for such noninfectious
problems.
 Optimal
therapy for pregnant women with active Lyme disease has not
been established. In early pregnancy the intravenous therapy
for serious central nervous system disease should be used.
In the third trimester, oral Amoxicillin, 1000 mg 3 or 4
times a day for 30 days is probably
adequate. Optimal
therapy for pregnant women with active Lyme disease has not
been established. In early pregnancy the intravenous therapy
for serious central nervous system disease should be used.
In the third trimester, oral Amoxicillin, 1000 mg 3 or 4
times a day for 30 days is probably
adequate.
 Corticosteroids
may be useful to supplement antibiotics in complete heart
block, facial palsies, and neuritis, and locally in chronic
arthritis. In severe or chronic arthritis and synovitis,
orthopedic surgery may be indicated. Corticosteroids
may be useful to supplement antibiotics in complete heart
block, facial palsies, and neuritis, and locally in chronic
arthritis. In severe or chronic arthritis and synovitis,
orthopedic surgery may be indicated.
Prevention
 A
vaccine to prevent Lyme disease was developed a few years
ago by SmithKline Beacham, and was approved for use in the
United States. The name of the vaccine was
LYMErix™. It was approved for persons
between ages 15 and 70 years. It was given in a series of
three doses, the second dose one month after the first, and
the third dose one year after the first dose. Maximum
immunity may not occur until after the third dose.
LYMErix™ will not prevent the disease in
persons who have unrecognized infection at the time of
vaccination; it is NOT a cure for Lyme disease. Protection
from other tick borne illnesses, such as babesiosis or
ehrlichiosis, does not result from treatment with
LYMErix™. SmithKline Beacham subsequently
merged to form GlaxoSmithKline Pharmaceutical Company. Sales
of the vaccine were low because of problems with degree of
immunity obtained and the delay of a year for full
effectiveness because of the need for three timed
injections. There were also law suits and reports of side
effects that may or may not have have been related the use
of the vaccine. Review by the Food and Drug Administration
resulted in no recommendation to discontinue its use.
However, GlaxoSmithKline, citing "cost effectiveness"
reasons, withdrew Lymerix™ vaccine from the market in
February 2002. While Lymerix vaccine is no longer marketed
here, there are other manufacturers. The Center for Disease
Control still has guidelines on Lyme vaccine at this link:
Lyme
Disease: Vaccine
Recommendations on
their website. A
vaccine to prevent Lyme disease was developed a few years
ago by SmithKline Beacham, and was approved for use in the
United States. The name of the vaccine was
LYMErix™. It was approved for persons
between ages 15 and 70 years. It was given in a series of
three doses, the second dose one month after the first, and
the third dose one year after the first dose. Maximum
immunity may not occur until after the third dose.
LYMErix™ will not prevent the disease in
persons who have unrecognized infection at the time of
vaccination; it is NOT a cure for Lyme disease. Protection
from other tick borne illnesses, such as babesiosis or
ehrlichiosis, does not result from treatment with
LYMErix™. SmithKline Beacham subsequently
merged to form GlaxoSmithKline Pharmaceutical Company. Sales
of the vaccine were low because of problems with degree of
immunity obtained and the delay of a year for full
effectiveness because of the need for three timed
injections. There were also law suits and reports of side
effects that may or may not have have been related the use
of the vaccine. Review by the Food and Drug Administration
resulted in no recommendation to discontinue its use.
However, GlaxoSmithKline, citing "cost effectiveness"
reasons, withdrew Lymerix™ vaccine from the market in
February 2002. While Lymerix vaccine is no longer marketed
here, there are other manufacturers. The Center for Disease
Control still has guidelines on Lyme vaccine at this link:
Lyme
Disease: Vaccine
Recommendations on
their website.
 Immunity
from the vaccine is not 100%, and the duration of immunity
that does develop is not well established. Previous
infection with Lyme disease does not necessarily result in
immunity, and reinfection is possible. Therefore, the
vaccine may also be useful in persons who have a history of
previous Lyme disease. Persons most at risk who might
benefit from the vaccine would seem to be those who work
outdoors, in tick-infested wooded and grassy areas.
Occupations that would appear to be at higher risk are
landscapers, foresters, wildlife and park managers, as well
as those who engage in hiking, camping, fishing and hunting
in such areas. Most cases of Lyme disease in the United
States are thought to be acquired in the peri-residential
environment, through routine activities of property
maintenance, recreation (including golf), and /or exercising
pets. Immunity
from the vaccine is not 100%, and the duration of immunity
that does develop is not well established. Previous
infection with Lyme disease does not necessarily result in
immunity, and reinfection is possible. Therefore, the
vaccine may also be useful in persons who have a history of
previous Lyme disease. Persons most at risk who might
benefit from the vaccine would seem to be those who work
outdoors, in tick-infested wooded and grassy areas.
Occupations that would appear to be at higher risk are
landscapers, foresters, wildlife and park managers, as well
as those who engage in hiking, camping, fishing and hunting
in such areas. Most cases of Lyme disease in the United
States are thought to be acquired in the peri-residential
environment, through routine activities of property
maintenance, recreation (including golf), and /or exercising
pets.
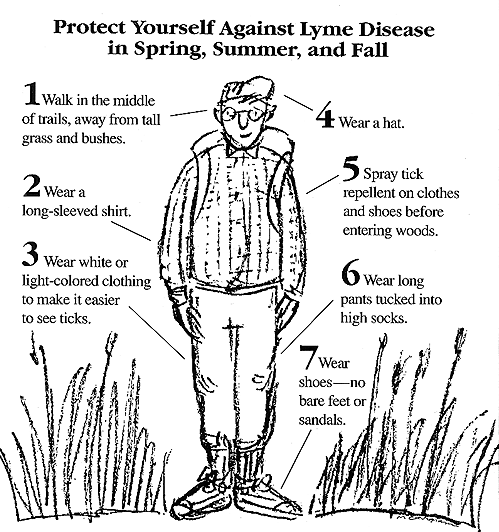
 Common
sense preventive methods include wearing long sleeved
shirts, long pants rather than shorts, tucking pants into
socks, treating clothing with tick repellant, and checking
for and removing attached ticks. Common
sense preventive methods include wearing long sleeved
shirts, long pants rather than shorts, tucking pants into
socks, treating clothing with tick repellant, and checking
for and removing attached ticks.
Bibliography
Centers for Disease
Control and Prevention. Lyme Disease -- United States,
1996. MMWR, June 13, 1997; Vol. 46:23;533-534
Duffy J. Lyme disease. Ann Allergy 1990 Jul;
65(1):1 - 13
Feder HM Jr., Gerber MA,
Krause PJ, Ryan R, and Shapiro ED. Early Lyme
disease: a flu-like illness without erythema migrans.
Pediatrics 1993 Feb; 91(2): 456-9.
Goldings EA, and Jericho
J. Lyme disease. Clin Rheum Dis 1986 Aug; 12(2):
343-67.
Morgan M, and Nathwani
D. Facial palsy and infection: the unfolding
story. Clin Infect Dis 1992 Jan: 14(1): 263 -
71.
Pachner AR, Duray P, and
Steere AC. Central nervous system manifestations of
Lyme disease. Arch Neurology 1989 Jul; 46(7): 790 -
5.
Satz N. Immunologie
und diagnostische Testverfahren bei der Lyme-Borreliose.
Schweiz Med Wochenschr 1991 Nov 21; 122(47): 1779 - 91.
(Published in German)
Sigal LH. Current
recommendations for the treatment of Lyme disease. Drugs
1992 May; 43(5): 683 - 99.
Slade JC and Johnson
RC. Lyme Disease. Chapter 166, Textbook of
Internal Medicine/Editor in chief, Kelly WN. Second
edition. J. B. Lippincott Company, Philadelphia. 1992. pp.
963 - 5.
Nowakowski J, Schwartz I,
Nadelman RB, et al. Culture confirmed infection and
reinfection with Borrelia burgdorferi. Ann Intern
Med 1997; 127:130-132.
Schwartz BS, Goldstein MC, Childs JE. Longitudinal
study of Borrelia burgdorferi infection in New Jersey
outdoor workers. 1988-1991. Am J Epidemiol,
1978;108(4):312-321
Schwartz BS, Goldstein MD. Lyme disease in outdoor
workers; risk factors, preventive measures, and tick removal
methods. Am J Epidemiol,
1990;131(5):877-885
Links
Lyme
Disease. a
public information guide from the Centers for Disease
Control and Prevention, National Center for Infectious
Diseases, Division of Vector-Borne Infectious Diseases,
Atlanta, Georgia 30333
American
College of Physicians Lyme Disease
Initiative
Page 17
|
